
How Life Uses Atoms
As we learned from Part 2 of this series, the ingredients for life are all around us. Some atoms, like nitrogen and oxygen, are abundant, so it comes as no surprise that living organisms have them in abundance in their bodies. Others, like boron and zinc, are less abundant so they have a more specialized purpose, which we will cover in this article.
It’s up to living organisms to take atoms of all shapes and sizes from their surroundings and put them inside their bodies in the appropriate quantities at the right time. While there are patterns to the atomic composition of all organisms, no two bodies are the same. In addition, different strategies have developed over time to help organisms acquire atoms. To me, one of the most interesting topics in biology is discovering all the different food sources that life on Earth takes advantage of to survive. To illustrate this point, let’s look at the atomic composition of living organisms, with an emphasis on plants.
The six most common atoms that make up living organisms on Earth are mnemonically known as CHNOPS (Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur). I remember it because it sounds like chin-ups. Plants fall under the category of living organisms, so they need CHNOPS in the highest amount. In fact, carbon, hydrogen, and oxygen make up a whopping 94% of a plant by weight. Nitrogen is next in line at 3%. Luckily for them, these four atoms are abundant in most natural systems, which means plants have access to 97% of their weight right off the bat. Yes, even nitrogen.
Carbon, Hydrogen, & Oxygen
Plants start the process by obtaining their carbon and oxygen as carbon dioxide (CO2), which they vacuum through the mouths in their leaves called stomata. Hydrogen is transported to the leaf in the form of root-absorbed water (H2O). What happens next is a feat nearing the miraculous.
Plants are able to strip electrons from extremely stable water molecules that don’t want to give them up. This causes water to unzip into two free hydrogen atoms and one free oxygen atom, as previously mentioned. Next, the hard-earned electrons are welded onto another stable compound, CO2, which doesn’t want them. These extra electrons attract the free hydrogen atoms from the splitting of water. The result is that plants have created their own food in the form of a Carbo-Hydrate (C+H2O). They then weld six of these sugars together into a compound known as glucose (C6H12O6). Dr. Brian Cox, British astrophysicist and professor of particle physics, writes in his book Forces of Nature, “This may seem unnecessarily complicated, but it probably isn’t. If you gave a chemical engineer the job of pulling electrons off water and putting them onto carbon dioxide, she’d probably laugh in your face.” And yet, this process of photosynthesis is exactly what plants do day in and day out to make the glucose they need to survive.
In summation, photosynthesis is responsible for capturing C, H, and O, which end up becoming 94% of the weight of a plant. These atoms become the raw material that is used to build the structure of the plant, as well as the energy source to power all the processes in a plant’s lifecycle (and eventually everything else!) by storing usable solar energy between carbon bonds.
The efficiency of photosynthesis is arguably the most important factor for plant health, plant production, and yield. As it turns out, photosynthetic efficiency is much lower than theoretical values predict, with one study from MIT showing that plants convert light to sugars at 6% efficiency.7 Further research shows yields can be increased by more than 40% for various crops that were genetically modified to have higher photosynthetic efficiency.8 Whether you believe genetic modification is the way to go or not is beside the point. The point is that glucose (C6H12O6) production is one of the lowest hanging fruits we can focus on to improve the health and productivity of plants.
This is why I personally dislike the concept that C, H, and O are not textbook “nutrients”. Even though a lot of other nutrients affect photosynthesis, and consequently C, H, and O levels in the plant, I still believe the heart and soul of the process is left out by excluding them. The tide is slowly changing, however. For example, the “Plant Health Pyramid“ developed by regenerative consultant John Kempf places improved photosynthetic efficiency at the base of the 4-part pyramid. Kempf teaches that plants with the capacity to produce more glucose and fuse a higher percentage of these glucoses together will be in a much higher state of health and productivity. Glucose production and fusing glucose both require the work of cellular helpers called enzymes. Enzymes fall under the category of protein, which leads us into the importance of the nitrogen atom.
developed by regenerative consultant John Kempf places improved photosynthetic efficiency at the base of the 4-part pyramid. Kempf teaches that plants with the capacity to produce more glucose and fuse a higher percentage of these glucoses together will be in a much higher state of health and productivity. Glucose production and fusing glucose both require the work of cellular helpers called enzymes. Enzymes fall under the category of protein, which leads us into the importance of the nitrogen atom.
Nitrogen
Proteins are among the most abundant molecules in the bodies of living organisms. While carbohydrates are fairly simple in that they form much of the structural skeleton and hold energy from the sun, proteins perform a near infinite number of jobs. In fact, one cell may contain hundreds or thousands of proteins that all have different jobs. These jobs may be to support the structure of the cell, help cells move, act as signals so cells can communicate, or facilitate the building or breaking of other molecules, which is what enzymes do.
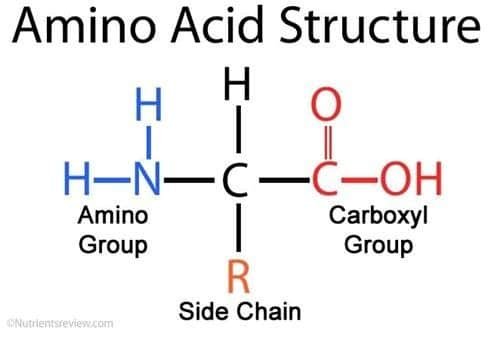
All proteins, no matter their job, are made up of units called amino acids that are linked together. Anytime you see “amine” or “amino”, think nitrogen. Every single amino acid contains a nitrogen atom, which is a big reason why it’s needed in such high quantities. The image below shows the basic structure of all amino acids: An amino group, a carboxyl group, and a side chain. The side chain is the part that can change, which gives rise to different amino acids. As it pertains to living creatures, roughly twenty amino acids are used to build proteins essential to biological creatures on Earth. If interested, follow this link to see a list of them.
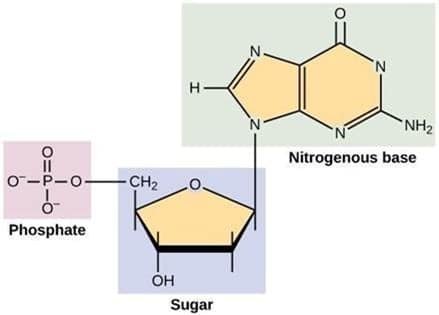
Nitrogen is also a key component of nucleic acids, the genetic compounds in living organisms. DNA (DeoxyriboNucleic Acid) is a nucleic acid which contains the blueprint for the production of proteins that make up living organisms. RNA (RiboNucleic Acid) is another nucleic acid, but its job is to transport and translate the information contained in DNA to sites that turn the blueprint into proteins.
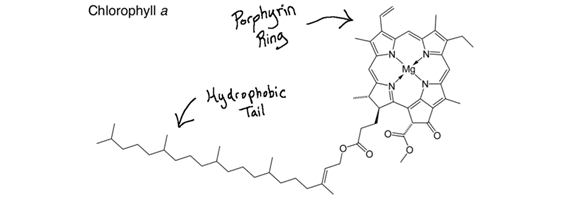
Lastly, nitrogen can be an important constituent of molecules that don’t fall under the umbrella of carbohydrates, proteins, nucleic acids, or fats. One such molecule is chlorophyll. Each green-reflecting chlorophyll molecule contains four nitrogen atoms, which is why plants often green-up with nitrogen fertilizer applications. More chlorophyll molecules are produced, which increases the amount of green light from sunlight reflected back to your eye. Basically, nitrogen is at the heart of almost every single process happening in living organisms.
The final 3% of atoms that make up the weight of living organisms originate from rocks and minerals in the Earth’s crust. Abiotic (non-living) or biotic (living) factors chisel them away and make them available for uptake by a microbe, plant, or animal. Once these atoms are released from minerals, they cycle in and out of living organisms as they live and die, get transported to locations via wind or water or become rock or minerals again by combining with other atoms.
Phosphorus
Phosphorus in the soil is notorious for quickly transforming back into a type of rock called apatite. (Fun fact: the hard outer surface of bones and teeth are made of calcium phosphate, a type of apatite.) This quick transformation back to rock happens to phosphorus that’s been chiseled away naturally and phosphorus that’s been applied as a fertilizer, with some estimates showing as little as 10–15% of phosphorus applied in fertilizers and manures is taken up by plants in the year of application.10 Despite this, phosphorus is a crucial atom for some of the most important processes in the bodies of living organisms. If you look at the image of DNA above, you will notice that phosphorus is the central atom for one of the basic units of DNA. The same goes for RNA.
In addition, phosphorus plays an integral role in a molecule called Adenosine Triphosphate (a.k.a. ATP), which is considered the energy currency for all of life. Whether we’re talking about microbes, plants, livestock, or humans, being alive and staying alive requires a lot of energy. ATP molecules are like mobile energy stations that can be transported to the site of an energy-intense process and release its energy to power said process.
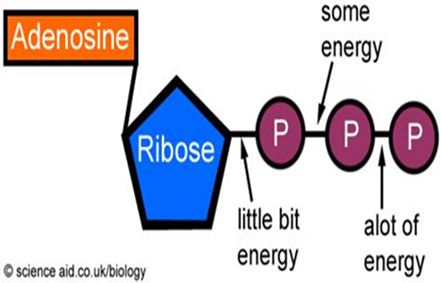
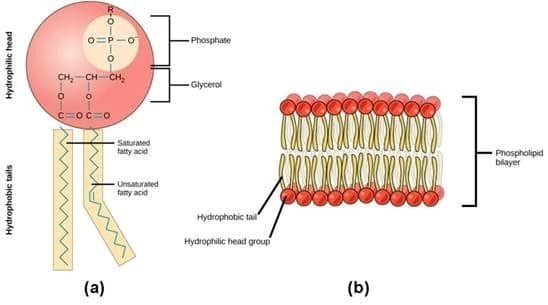
Lastly, phosphorus is an important component of the cell membranes of organisms. Cell membranes separate the cell from the surrounding environment, provide protection, and allow transportation of items in and out of the cell, among many other jobs. It’s hard to overstate the importance of phosphorus.
Sulfur
Rounding out the top six list of common atoms in a living organism is sulfur. This atom plays a central role in the proper functioning of proteins, as it allows the chain of amino acids to fold into the correct shape. In biology, function follows structure, so if a protein doesn’t have the right shape, it won’t work like it’s supposed to.
Two important amino acids contain sulfur in their side chains: cysteine and methionine. Sulfur in cysteine will bond with sulfur from another cysteine to form “disulfide bridges”, which folds the protein in the appropriate spots. This can make the difference between an active protein and an inactive one, as the image below depicts.
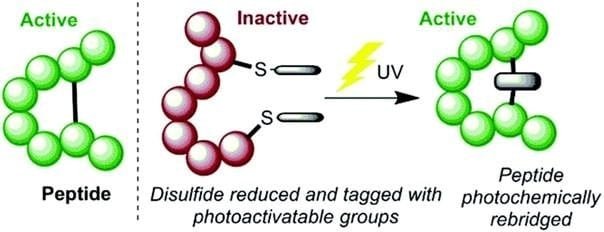
Methionine is also important as it is found in many proteins that uphold the structural integrity of the cell. Interestingly, sulfur is released during the burning of coal and other fossil fuels because these are old pieces of organic material whose sulfur is retained in their cells over time. Coal formed near brackish or saltwater environments contains higher levels of sulfur.11 Burning these ancient cells releases the sulfur as sulfur dioxide (SO2). Many farmers and ranchers used to receive annual sulfur amendments as the sulfur dioxide emissions dropped onto their land. Sulfur scrubbers and other environmental laws over the past decades have decreased the amount of sulfur dioxide in the air, and, consequently, the amount of free sulfur provided to the soil.
Bringing CHNOPS Together
To recap, atoms are the basic building blocks of life. They function like Legos by connecting with other atoms to make complex molecules that can do different tasks. Bonds are formed by the giving, taking or sharing of negatively-charged electrons that orbit around the positively-charged nucleus. The movement of electrons is what makes life happen, essentially. Due to the number of electrons orbiting their nuclei, carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur (CHNOPS) atoms make up the vast majority of a plant, or any other living organism for that matter. They are the main constituents of the main molecules: carbohydrates (C,H,O), proteins (C, H, N, O, S), fats (C,H,O) and nucleic acids (C, H, N, O, P). Notice that carbon, hydrogen and oxygen are found in all four groups, which illustrates the incredible importance of these atoms to living organisms. Carbon, hydrogen, nitrogen, and oxygen are found in the air and water prior to making up the body of a living organism. All other atoms originate from rocks or minerals in the Earth’s crust, including phosphorus and sulfur. Some atoms, like sulfur, can be found in gaseous forms in addition to mineral forms, but they originate from the mineral portion.
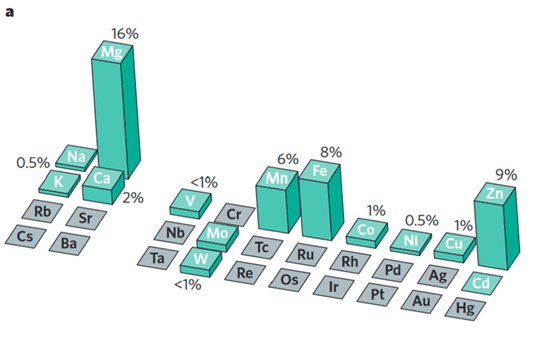
Metals: Best of the Rest
One interesting fact about atoms (a.k.a. elements) is that most are metals. Incredibly, CHNOPS are not, as you can see in the periodic table of elements below. Is it a coincidence they make up the vast majority of living organisms? I think not. Their natural abundance and nonmetal characteristics make them the perfect candidates to create stable molecules, kind of like the skeleton of a house. Now, it’s the job of the metal elements to make the house a home with their unique abilities. Metals currently recognized as essential to living organisms are Potassium (K), Calcium (Ca), Magnesium (Mg), Zinc (Zn), Copper (Cu), Manganese (Mn), Iron (Fe), Boron (B), Molybdenum (Mo), and Nickel (Ni). Some sources also list Silicon (Si), Sodium (Na), and Vanadium (V). Boron and silicon are technically “metalloids”, meaning they sort of behave like metals and sort of don’t. Chlorine (Cl) is the only essential atom after CHNOPS that is a nonmetal. However, it is still important, as chlorine is needed to break apart water molecules during photosynthesis and it aids in the operation of plant mouths (the aforementioned stomata).
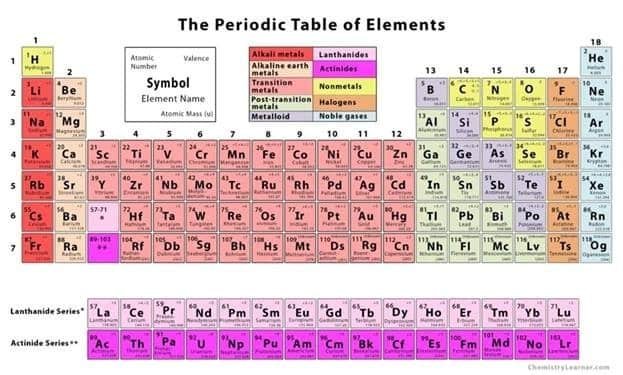
So, what makes an element a metal? Physical properties of metals include being shiny, conducting heat and electricity well, having a high melting point, and being malleable, among others. These are tied closely to the chemical properties of metals. (Remember, Chemical = Chemistry, and chemistry is simply the movement of electrons.) These chemical properties include the fact that metals have 1-3 electrons in their outermost shells, they lose these electrons readily and they all react with oxygen to form oxides (think oxidized iron a.k.a. rust).
The ability for metals to conduct electricity and move electrons is extremely important for living organisms. We wouldn’t be alive without them. Think of batteries in the TV remote or in the smoke alarm. Batteries power devices because there is a gradient of electrons that flow from high concentration to low concentration. As it turns out, living organisms utilize the flow of electrons as well, in what is called bioenergetics or bioelectricity (bio- means life or living things). For example, the nervous system of animals utilizes sodium and potassium atoms to create millivolt-level electrical impulses sent from the brain to the body and back. However, neurons aren’t required for bioelectricity, as the movement of sodium, calcium, and potassium atoms gives our heart muscle fibers the gradient for an electric spark it needs to beat. EEGs are simply recordings of the electrical signals in the brain, while EKGs are recordings of the electrical signals in the heart. These neuronal and non-neuronal bioelectric signals are utilized by all living organisms on God’s green Earth, not just humans and animals. In fact, scientists have studied plant bioelectric signaling for over 100 years now.12
The point is that metals greatly influence the movement of electrons, and electron flow is necessary for the use and production of energy in living organisms. In the house analogy, metals are like the electric wiring in the walls and batteries in appliances that allow countless processes to be done. Copper, nickel, manganese, and zinc are common atoms used in wires and batteries that living organisms also utilize to move electrons and conduct electricity in their bodies. Pretty cool, isn’t it?
Proteins, being the workhorses that perform the near infinite number of tasks required to keep living organisms functioning, are especially good at pairing with metals. To illustrate this, let’s look at one type of protein previously mentioned: the enzyme. Enzymes enable chemical reactions to happen, basically. Life-sustaining reactions that need molecules broken apart or fused together can take years, decades, or even centuries to happen on their own. Enzymes enable these reactions to happen in seconds, and almost half of all enzymes require the presence of a metal to function properly (you’re likely to see this metal called a “cofactor”).13
Nitrogenase is one such enzyme. (Anytime you see –‘ase’ at the end of a word, think enzyme). Anaerobic microbes, like rhizobia and azotobacter, deploy nitrogenase to break apart N2 molecules from the air and transform it into ammonia (NH3). Even though carbon, hydrogen, and oxygen make up the majority of the nitrogenase enzyme, it will not be able to function without a tiny amount of iron and molybdenum atomic batteries. Transformations all throughout the Nitrogen Cycle require enzymes at each step, such as ammonia monooxygenase, which requires copper and iron atoms, as well as hydroxylamine oxidoreductase which requires iron atoms as well.
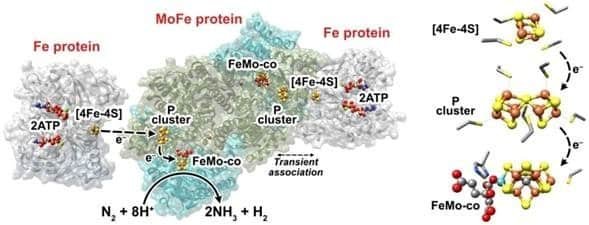
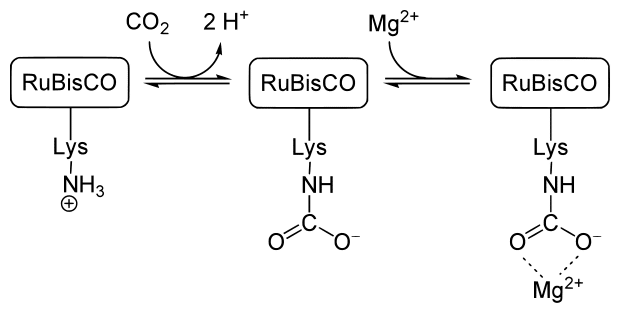
Inside of the plant, rubisco (Ribulose-1,5-bisphosphate carboxylase/oxygenase) is an important enzyme which catalyzes the first step in the fixation of carbon dioxide into glucose.14 Rubisco is the most abundant protein and enzyme on the planet. Its very presence allows for life on Earth to continue, and it won’t work without magnesium atoms. Chlorophyll molecules, while not enzymes, also require a magnesium atom to function, as you can see in the “Chlorophyll a” image a few paragraphs up.
Other important uses for metals include a group of one of the most abundant proteins in biology called “Zinc fingers” (Honestly!), antioxidant enzymes dependent on manganese, and the various uses of potassium and calcium, among an infinitely long list of others. Novel biochemical discoveries are coming out nearly every day on the topic of molecular structures and their functions.
The goal of this section was to get across that the quantity of a given atom in a living organism doesn’t equate to importance, because importance is subjective. Priority has been given to plant growth and fruit yield in the past hundred years or so, which is one reason why N, P, and K became top dogs. Their applications create visible plant responses in the ways we’ve decided are most important. Scientists, farmers, and ranchers did their best with the information they had, but we have the knowledge and the data today to prove that there’s much more to the story.
Different types of atoms have different jobs that work together not only for growth, but for the health and longevity of the microbe, plant, or animal. Keep this in mind when you further your education concerning the “macronutrients” and “micronutrients”. Another point worth noting is that the list of essential nutrients has grown over the decades, so it’s highly, highly unlikely that we have reached the pinnacle of understanding plant nutrition. In my humble opinion, we should view the current list as a work-in-progress, not gospel truth. Therefore, an updated definition of essential nutrients may do a lot of good. UC Davis professor Patrick Brown and collaborators provide such an updated definition, which reads, “A mineral plant nutrient is an element which is essential or beneficial for plant growth and development or for the quality attributes of the plant or harvested product, of a given plant species, grown in its natural or cultivated environment. A plant nutrient may be considered essential if the life cycle of a diversity of plant species cannot be completed in the absence of the element. A plant nutrient may be considered beneficial if it does not meet the criteria of essentiality, but can be shown to benefit plant growth and development or the quality attributes of a plant or its harvested product.”15 It’s a step in the right direction, if nothing else.
What’s Coming Next
Part 4 of this series dives into the dynamic world of nutrient cycling, revealing how elements constantly shift between living and non-living forms as they move through soil, plants, microbes, and the broader environment. By exploring concepts like mineralization, immobilization, and the surprising ability of plants to absorb organic compounds, you’ll see the soil as a living, active system rather than a passive storage medium with the goal of reframing nutrient management as a process of working with nature. As farmers and ranchers, we can use this knowledge to leverage biological cycles to improve efficiency, reduce inputs, and build healthier, more resilient growing systems.
Understanding how biology and nutrient cycling work together changes the way we approach soil fertility, plant health, and farm productivity. Regenerative management is about working with these natural processes to build healthier, more resilient systems. At Understanding Ag, our consultants help farmers and ranchers apply these principles in practical, profitable ways that improve efficiency, reduce input costs, and strengthen the long-term health of the land. To learn how these concepts can work on your farm or ranch, connect with an Understanding Ag consultant.
References
8https://academic.oup.com/jxb/article/70/4/1119/5322156?login=false
9https://www.advancingecoag.com/plant-health-pyramid
10https://www.amazon.com/Nature-Properties-Soils-15th/dp/0133254488 (pg 644)
11https://www.ucsusa.org/resources/how-coal-works#sources
12https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-3040.2006.01614.x
13https://www.whoi.edu/cms/files/WaldronRobinsonnature2009_53167.pdf
14https://www.annualreviews.org/doi/10.1146/annurev-biochem-040320-101244
15https://link.springer.com/article/10.1007/s11104-021-05171-w
The post 4 Ecosystem Processes: Nutrient Cycle Part 3 appeared first on Understanding Ag.















