
Where the Wild Atoms Are
Learning the story of nutrient cycling is a necessary first step to memorizing the nutrients and how to manage them appropriately. Most of us were “taught” the latter without the former, which is an extremely difficult way for our brains to remember something. The story provides the substance for the facts to cling onto in our minds. I challenge you to sit down and read a bullet-pointed list of facts on a topic you don’t know much about and try to recite the long list of facts you read once you’re done. This is a boring and ineffective way to learn something, but that’s the method we use to teach the rich topic of nutrition. Worst of all, this method fails to emphasize the fact that you are the leading character in the unfinished story of nutrient cycling that starts on the farm or ranch and ends up in the cells of human or animal consumers. Your management decisions dictate how the story is written. This may seem daunting, but remember, there are billions to trillions of supporting characters in your soil, air, and water who are chomping at the bit to help you write the story.
The Goal of Life: Moving Atoms
One of the main goals of living organisms is to bring specific atoms in specific quantities from outside of their body to inside their body in a form they can use. Many atoms are then sent to become one of four basic compounds, or “macromolecules”: carbohydrates, proteins, fats, and nucleic acids. Each will be described throughout the article. Because we don’t live in the vacuum of space, all of us living creatures on Earth are surrounded by atoms in the ground, water, air, and bodies of other living organisms. Let’s start with rock and move our way up.
Life Begins in the Rocks
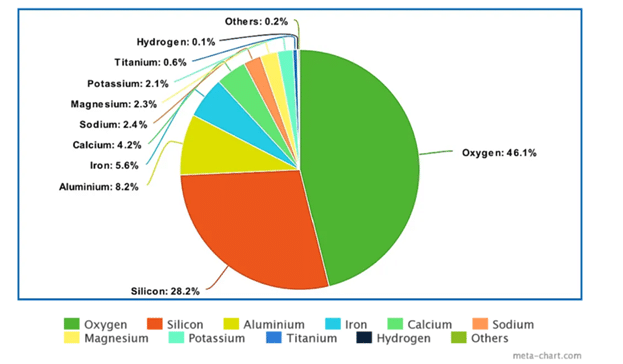
Earth’s crust is the outermost solid layer and, believe it or not, is made up mostly of oxygen, followed by silicon, aluminum, and iron. Most of us only think of oxygen as a gas, but oxygen combined with other atoms can be in a solid, liquid, or gaseous form. The size and number of electrons in an oxygen atom make it quite the promiscuous character, as it wants to bond with various other atoms in the vicinity. In the crust, oxygen bonds create solids, such as with silicon dioxide (SiO2), the main constituent of sand. In fact, oxygen readily bonds with many metals and semimetals, not just silicon.
Unlocking Nutrients: Biology vs. Time
Atoms in rocks and minerals are held too tightly for living organisms to utilize until factors chisel atoms away from each other. These factors can be non-living (“abiotic”) or living factors (“biotic”). One common biotic method of extraction is the secretion of strong chemicals by microbes to dissolve rock into its constituent atoms for the purpose of consuming them and inserting them into their body. Pretty darn cool, if you ask me. This process can happen on the timescale of hours, greatly accelerating the cycling of atoms out of rock compared to abiotic factors that may take hundreds to thousands of years.
The Role of Soil Biology in Nutrient Availability
The more biological activity in the soil, the more these atoms get extracted and can be used by living organisms. As you can see from the pie graph below, atoms important to plant growth, such as iron, calcium, magnesium, and potassium, are found in the rocks and minerals of the Earth in significant quantities.
Later, we will discuss the importance of the Total Nutrient Digestion (TND) soil test, which gives an idea of the full amount of various nutrients in the soil. Land managers are often surprised to learn that they have hundreds or even thousands of pounds of these nutrients in their soil. The challenge is to get this giant bucket of unavailable nutrients chiseled away and in the bodies of living organisms.
Water: The Universal Transport System
Oxygen also makes up a significant portion of the water that surrounds us, as it is one-third of the atoms and 90% of the weight of a water molecule (H2O; Two Hydrogen atoms and one Oxygen atom). Water is important to the Nutrient Cycle because it is considered the universal solvent, meaning it will magnetically bind with charged regions of other individual atoms or larger molecules.
Water can also break apart molecules into smaller pieces and carry them away, such as when table salt (NaCl) dissolves into a positively charged sodium atom (Na+) and a negatively charged chloride atom (Cl-). Similarly, water in the soil bonds with charged compounds and atoms that magnetically attach to water. This is important because plants can take up “water-soluble” nutrients like nitrate (NO3-) and ammonium (NH4+) through their roots. What water holds on to varies temporally and spatially. For example, hard water has a high concentration of calcium and magnesium dissolved in it, while purer water has much lower concentrations. Just like minerals build up on cups and silverware from hard water, soils with consistently high water tables result in a buildup of atoms, like sodium, left in the soil after water evaporates into the air.


Up in the Air
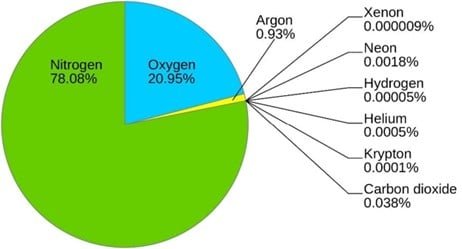
Moving upward, nitrogen makes up 78% of the air we breathe. We are literally bathing in nitrogen. In fact, over 30,000 tons (60,000,000 lbs) of nitrogen are above one acre at any time.1 Ironically, nitrogen is often the atom that limits plant growth and decreases yield potential. This is because nitrogen in the air is not available to living organisms, like the atoms held tightly in rocks and minerals.
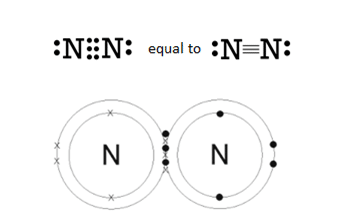
To understand why, recall that one nitrogen atom contains five electrons in its outermost shell, which means it wants three more to reach the magical, stable number of eight. As it turns out, two nitrogen atoms can share three of their own electrons to give both nitrogen atoms eight in their outer shell, making N2 gas. Remember, an electron shared between atoms counts for both atoms. This process creates a triple-bond between the two nitrogen atoms which requires an enormous amount of energy to shift electrons around and break it apart. Miraculously, many living organisms have devised strategies to gain energy from the separation of two nitrogen atoms. Legume-associated rhizobia bacteria are one such species. We will dig deeper into this process later.
Oxygen and the Energy of Life
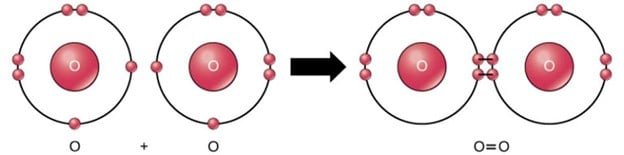
While oxygen is the most important atom in the air for us aerobes, it is a distant second in terms of abundance. The amount of oxygen atoms in the air rise or fall based on the quantity of photosynthesizing organisms on land and in the water. Photosynthesis splits water (H2O) into 2 hydrogen atoms and one oxygen atom. As you may recall, oxygen is promiscuous and doesn’t like to be alone. One oxygen atom has six electrons in its outer shell, meaning it wants two more to reach the magical, stable number of eight. Inside the plant, two oxygen atoms bump into each other and share two of their electrons in what’s called a double bond. This creates O2 gas in the air. The double bond of O2 requires less energy to break compared to the triple bond of N2 gas, just like a door with two locks is easier to open than a door with three locks. Air breathers like us take advantage of this fact and crack open O2 to use the energy in our food to stay alive. This is called aerobic respiration and it’s the reason why animals, plants, fungi, bacteria, and all aerobic organisms bring oxygen inside their bodies.
What’s Coming Next
In the next installment, we’ll learn the importance of CHNOPS (Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur) and how these six elements are acquired, transformed, and used within living organisms, especially plants. We’ll also uncover how photosynthesis produces glucose, the primary energy source and structural building block of life. And finally, we will explore the critical roles of nitrogen in proteins and DNA, phosphorus in energy transfer (ATP) and cell structure, sulfur in protein function, and much more.
Bringing It Back to Your Operation
Understanding how nutrients move through soil, water, air, and living organisms isn’t just science—it directly shapes your management decisions and long-term profitability. Every choice you make influences how effectively these cycles function, and when managed well, biology does much of the work for you. If you’re interested in applying these principles on your farm or ranch, reach out to an Understanding Ag consultant to turn these concepts into practical, profitable strategies tailored to your operation.
References
1https://2019.fertilizerreport.org/wp-content/uploads/2020/02/TFI-NutriFacts-Nitrogen_v2.pdf
The post 4 Ecosystem Processes: Nutrient Cycle (Part 2) appeared first on Understanding Ag.















